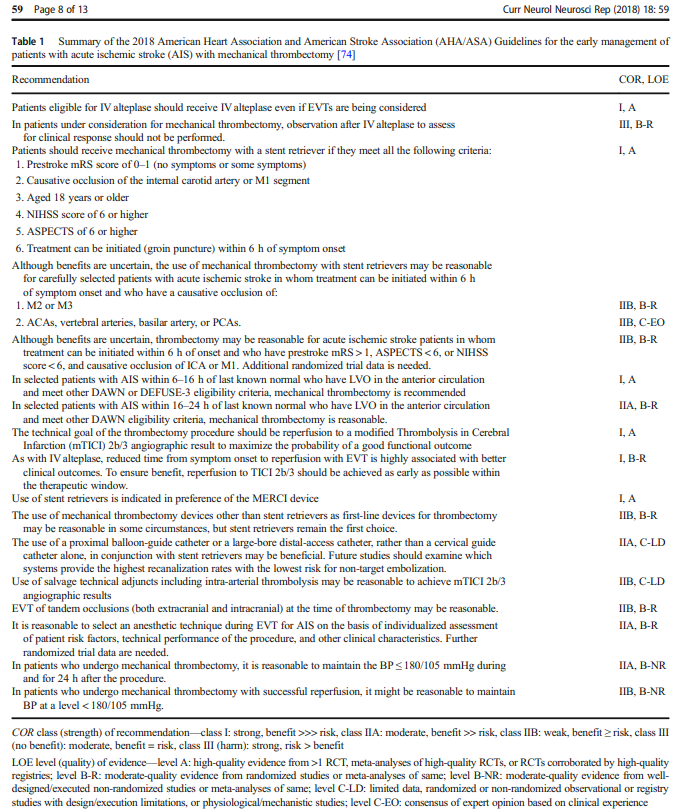
“Moderate consumption of unsweetened or sugar-sweetened coffee was associated with lower mortality in an observational study.“
Comment:
“Associations between coffee consumption and mortality surely are confounded by numerous factors. If there is really a causal protective link, it might be related to coffee’s chlorogenic acids, which — in addition to caffeine — have an antioxidant effect and inhibit platelet aggregation, as an editorialist notes. A study limitation is that, on average, only 4 g of sugar were added to sweetened coffee (likely less than many Americans add). Nevertheless, this study reaffirms that even sweetened coffee is likely not harmful and might even be beneficial.”
Dressler DD. Coffee and Mortality: Sweetening the Pot NEJM Journal Watch. (June 16, 2022)
Citations:
Liu D et al. Association of sugar-sweetened, artificially sweetened, and unsweetened coffee consumption with all-cause and cause-specific mortality: A large prospective cohort study. Ann Intern Med 2022 May 31
Wee CC. The potential health benefit of coffee: Does a spoonful of sugar make it all go away? Ann Intern Med 2022 May 31
_____________________________________
“Two studies confirm an association between coffee intake and lower risk for death in diverse populations.”
Comment:
“These studies provide support that moderate coffee intake (2–5 cups daily) is safe and associated with reduced risk for death, probably in a dose-dependent fashion. If you’re still debating the health benefits of coffee for your patients, you may want to do it over a cup of joe.”
Dressler DD. Moderate Coffee Consumption and Mortality: Good News NEJM Journal Watch. (July 11, 2017)
Citations
Park S-Y et al. Association of coffee consumption with total and cause-specific mortality among nonwhite populations. Ann Intern Med 2017 Jul 11; [e-pub].
Gunter MJ et al. Coffee drinking and mortality in 10 European countries: A multinational cohort study. Ann Intern Med 2017 Jul 11; [e-pub].
Guallar E et al. Moderate coffee intake can be part of a healthy diet. Ann Intern Med 2017 Jul 11; [e-pub].
_____________________________